Archive for the ‘Surgery’ Category
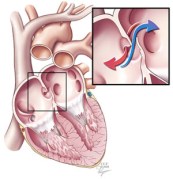
A patent foramen ovale is a defect in the wall between the two sides of the heart that allows the passage of blood and its contents. This course bypasses the lungs. (Courtesy Cleveland Clinic)
One controversial cause of some strokes is a small hole between the two sides of the heart known as a patent foramen ovale. Although rarely symptomatic for patients, this hole allows blood clots that occur in otherwise healthy individuals to bypass the lungs and lodge in critical arteries that serve the brain. In individuals without this defect, these small blood clots would normally lodge in the small vessels of the lungs and typically never lead to a disease state (see note at end for a more complete explanation). Because of the unresolved structural defect, conventional thinking considers these individuals at increased risk for future strokes that remains even after typical stroke prevention strategies.
Kurt Amplatz is an interventional radiologist who has spent his career developing a number of devices to repair these patent foramen ovale and other defects using a catheter-based device that closes the hole with a permanent metal disc. The procedure is similar to cardiac catheterization procedures used for patients with coronary artery disease (e.g., heart attacks).

An Amplatzer occluder deployed on the end of a catheter. (Courtesy St. Jude Medical)
Two recent papers(1,2) in the New England Journal of Medicine report findings from long-term studies designed to demonstrate the expected benefit of using these “Amplatzer” devices versus traditional medical therapies (e.,g., aspirin, Coumadin®, Plavix®). The Journal effectively uses these two studies to demonstrate just how fine a line of improvement may be found with use of the devices and ultimately concludes that true believers and skeptics will likely not be swayed from their opinions by the limited findings of both studies.
What I found most interesting about this recent revival of the debate of catheter-based occlusion devices is the near-zero discussion of the cost component of these devices. The Amplatzer occluder used in these studies was not a one-time quick-fix for these patients but was a supplemental therapy that was often used in conjunction with traditional medical therapies. Although exact pricing is not available, the cost of the device alone adds an additional $3,000-5,000 to the cost of the patient’s care and insurance is usually billed an additional $10,000-$25,000 for the procedure.
With the current evidence, these devices add additional costs and procedural risks to a patient’s care without demonstrating definitive benefit. Addressing the escalating cost-problem in U.S. healthcare starts with regulatory authorities scrutinizing care scenarios such as this one to determine if we are getting value for money in procedural medicine.
Note on blood clots: Venous thromboembolisms are a major cause of morbidity and mortality. However, the specific sequence of events that produces small clots that the human body can easily degrade versus those that cause life-threatening events is poorly understood. Although any blood clot seen in the healthcare setting is typically treated as if it were potentially life-threatening, the general thinking is that small blood clots as in the example given above are somewhat routine in the older population and can resolve spontaneously if not symptomatic.
A number of recent academic studies have shown that global health experiences are becoming more essential in the eyes of medical trainees. One particular study suggested that the vast majority of current surgical residents are interested in global health experiences. Our research group at Emory has further shown that medical students are likely considering global health offerings when students evaluate residency programs. The reality for residency programs today is that failing to offer opportunities in global health may be harming their ability to recruit the best applicants for their program. However, medical schools and residency programs alike have had difficulty overcoming the logistical difficulties while also maintaining the quality of medical education provided during such experiences.

Drs. Jahnavi Srinivasan and Viraj Master demonstrate point-of-care ultrasound techniques to medical students Lee Hugar and Pete Creighton during Emory Medishare’s surgical camp in Hinche, Haiti.
For the last five years, I have worked intimately with a small group of faculty and students at Emory University School of Medicine to design a for-credit international surgery elective that attempts to demonstrate the feasibility of such a training experience for medical student. A long-form retrospective piece on the effort and how it has evolved to meet the needs of multiple stakeholders has just been published in the Bulletin of the American College of Surgeons. The key takeaway from our group’s experience is that the common criticisms of these short-term trips fail to wholly encompass the range of benefits being provided. If one assesses solely the educational value or exclusively the burden disease effect for the patient population, a perspective that incorporates the cumulative benefit of these programs is lost.
As I have started the transition from medical student to general surgery residency, it has become increasingly important for me to find a way to communicate our message to the next generation of medical students. At Emory, I have no doubt that an exceptional class of rising senior medical students with global health experiences will have no problem continuing to build on the model there. But what can be done for other medical schools that don’t have such a program or have not operationalized it in a manner that can continue across multiple years? After considerable thought and planning, we have released an early version of a website, www.MedStudentTrips.org, that will serve as a repository of public clinical manuals, planning documents, and advice for those looking to replicate the Emory Medishare model at their own school. In passing on such knowledge, I hope to catalyze such efforts at other institutions in the future.
For information on Emory Medishare, the student-faculty medical humanitarian collaborative discussed in this article at http://www.emorymedishare.org.
For those looking to design such a program at their own medical institution, Emory Medishare has posted many of its public resources at http://www.MedStudentTrips.org.
Editor’s Note: A change in the publication schedule has been made for the month of November. This post below replaces a previous November post on pharmaceutical markets in developing countries that will be re-published in the future.
Disclaimer: Every pregnancy is unique. The below discussion focuses on population-wide assessments of effectiveness and should not be used to make a decision about one’s personal healthcare. Any questions about the issues raised below with regard to the reader’s own pregnancy should be discussed with their obstetrician.
When a pregnant woman arrives to a hospital in labor, she will typically be whisked away to a Labor and Delivery ward. For those familiar with birth, the timing of what follows is highly unpredictable. If in true labor, she will be whisked to the room where she will deliver, and be allowed to progress for the often hours-long wait prior to delivery. After being immediately assessed, most institutions include protocols that have the ward nurses place a continuous fetal heart rate and uterine contraction monitor. The information gathered by the probes is then usually electronically transmitted to a central server for display on any number of desktop monitors and overhead displays scattered throughout the ward. The principal behind this monitoring is for the medical team to be able to readily assess the basic vital signs of labor on any patient from any location in the ward. For example, fetal heart rate decelerations are one recognized sign of fetal distress, and many institutions train staff to immediately respond to such a feature observed on monitoring. A frequent result of “ominous” or “nonreassuring” fetal heart rate patterns is that further observation of the fetal heart rate does not improve the assessment of fetal health and an emergent Caesarean section ensues.

As comforting as continuous fetal monitoring may appear, the devices’ tendency to prematurely warn of fetal distress may actually worsen outcomes for mother and baby. (Photo Courtesy: Medgadget)
Conventional wisdom would suggest that the current widespread distribution of such telemetry monitoring for active labor has saved the lives of countless women and their progeny. Until the twentieth century, pregnancy was one of the most dangerous periods of a woman’s life, and much of this was the result of difficulties during active labor without the ability to safely remove the near-term pregnancy. Even today, Caesarean section – with only minor technical modifications in the ensuing period – remains the standard of care for achieving optimal outcomes when persistent fetal distress is detected during active labor. Logically, one would then think that real-time vitals of the fetus would be the ideal technological assistance for fetal health surveillance during this period of pregnancy.
Although such reasoning appears sound and is routinely used by purveyors of continuous heart rate monitoring, the scientific evidence to date does not support the use of such devices. Although proponents of continuous fetal monitoring have cited a large recent study that suggests otherwise, the best scientific evidence suggests that continuous fetal heart rate monitoring results in the same mortality outcomes for mother and fetus as what can be achieved with the traditional form of fetal health monitoring, or “intermittent auscultation” (a nurse rounding on the mother every 15 minutes and listening for the fetal heart rate with a stethoscope-like device). Worse, not only are mortality outcomes unchanged by an expensive piece of hospital IT infrastructure, but the studies have also shown that the number of Caesarean sections increases with the use of continuous fetal heart rate monitoring. What this constellation of findings suggests is that such monitoring does not improve health outcomes but also has the unfortunate disadvantage of “sounding the alarm” too early. In other words, continuous monitoring appears to be picking up fetal distress that is transient that may be more accurately described as “fetal discomfort.” These findings suggest that if such cases were left alone, the distress would often resolve on its own without any lasting effect on health outcomes for mother or baby.
To summarize, the amassed evidence to date suggests that continuous fetal monitoring does not improve newborn mortality and increases the number of C-sections performed when it is used. Why then is it the standard of care across much of the U.S.? I’ve put exactly this question to a number of my mentors and colleagues, and the answer is invariably the same. Evidence aside, no jury in the United States is going to be kind to the obstetrician who goes against the tide of medical practice to adhere to what “stuffy” academics suggest is a more effective treatment strategy. That a minority of institutions in the U.S. even still allow practitioners to use intermittent auscultation is testament to the American College of Obstetrics and Gynecology’s measured acceptance of both forms of fetal monitoring.
What troubles me the most about the state of fetal monitoring today is the potentially massive quality transformation that exists and the limited work being done to explore this issue further. Labor is one of the most common conditions requiring hospitalization and the question of how it is monitored affects every single one of those admissions. Definitive research is needed to settle this debate. Because such research may very well prove the futility of a profitable medical technology, it is likely that professional advocacy is the only way such work will get the necessary funding to conduct a large randomized trial. Without it, we will continue to perform a practice that is contributing to what may be thousands of unnecessary surgeries a year and a considerable cost burden to our healthcare system.
Disclosure: I have no financial or professional interests related to continuous fetal heart rate monitoring or other matters discussed above.
For those of you familiar with my background and specific research interests in global health and quality improvement, it should be no surprise that I routinely get asked how do I connect two very disparate fields of medicine like global health and quality improvement. Just last week, a fellow of my college at Oxford, asked for further clarification. I still find myself falling into the trap of assuming the overlap between the two is obvious to others since that overlap is where most of academic work currently focuses. This particular fellow was a healthcare economist with an interest in financial crises and their impact on global health, so I incorrectly assumed that he would “get it.” This lack of discernment is unusually common, and I believe it only reinforces the relative lack of interest even within the global health establishment for the issues that I find most engaging.
Before one can understand my area of overlap between these two fields, it is important that I re-frame what these terms mean. To understand how I use the terms and my particular interests within each, most would benefit by briefly reviewing prior posts on the two (global health, quality improvement). At the macro-level, I find both of fields fascinating and ultimately the causative factor for why I am pursuing a career in academic medicine (i.e., not private practice). For me, these two fields are the current focus of healthcare’s greatest obstacles. Globally, we have billions of people around the world who cannot access even basic healthcare services aligned to 21st century standards of clinical care. In the industrialized world, we find that most countries are unable to provide healthcare services in a manner that maximizes capabilities given a set of constrained financial resources.
Albeit in different contexts, the kinds of systematic and institutional inefficiencies that ultimately impair the delivery of quality healthcare in modern health systems like the U.S. are also evident in the delivery of healthcare in even the most resource-limited environments. Global health is quickly reaching a point where the technical capabilities needed to address the world’s healthcare problems (e.g., effective antibiotics, vaccine development and production technologies, low-cost anesthesia equipment) are available, but global health interventions often lack the operational competence needed to achieve their goals.
My belief and where much of my research efforts are focused is that we can use the quality improvement frameworks being developed for modern healthcare systems to also improve the healthcare delivery in less developed healthcare settings as well. Global surgery is an ideal area in which to adopt these analytic models because of the process-driven nature of the surgical resource procurement (e.g., anesthesia and surgical equipment, pharmaceuticals for infection prevention and anesthesia) and direct patient care.

Outside Hôpital St. Thérèse (Hinche, Haiti)
“Global surgery” is a relatively new addition to its parent field of global health. Although the notion of surgeons from the industrialized world sharing skills and equipment with resource-scarce environments has been around for decades (largely arising out of the medical operations of the world powers’ militaries), “global surgery” as an academic interest within traditional global health circles has emerged only recently. The increasing interest in the global burden of surgical disease has largely paralleled the relatively new awareness of noncommunicable disease as a global problem.
I should be clear that a consensus has only just begun to form in the last 10 years around the broadening of the definition of global health. For example, although there is an increasing effort to use “global health” in a more appropriate, literal sense, many well-qualified academics still associate the term with the unique disease burden of low- and middle-income countries. Say “global health” in a professional healthcare setting and poor, non-white people suffering from malaria or HIV is what the majority of the audience will be envisioning. It is only recently that the more complex problems of noncommunicable diseases like diabetes and cancer that span the industrialized and developing world have been authentically engaged by the global health community.

Inside Hôpital St. Thérèse (Hinche, Haiti)
Within global surgery, I am most interested in the persistent difficulty we have of finding the means of providing even the most basic surgical care for all the world’s communities. Although it will be decades before the poorest countries will have the means of performing complex surgeries like spinal fusions or coronary artery bypasses, the infrastructure and skills necessary to perform hernia repairs and thyroidectomies is well within reach. These latter cases may be less “newsworthy” but they represent substantial quality-of-life improvements for patients suffering from surgically-correctable disease. Although the field of global surgery is still in its nascent stages, a number of expert bodies are catalyzing around the idea of essential surgical care (e.g., ASAP Today, WHO GIEESC) and how best to deliver it.
A note on the pictures: The pictures in this post include two from a humanitarian surgical mission I joined in June 2009. This was the first of a series of many missions (more in a later blog post). I inserted these two images here to demonstrate how even the limited infrastructure of Hôpital St. Thérèse (see “outside” photo) can provide the platform for delivering basic surgical care (see “inside” photo). The bottom picture is provided courtesy of Nick Vittone of AtlantaAperature.com.
A man visits a surgeon about a lipoma (usually a benign glob of fat) on his arm that has been aesthetically bothering the patient for a number of years. The surgeon assesses the lipoma and rules out any concerning other health problems. The man is booked for surgery three weeks later. The patient arrives on-time to an outpatient surgery center for the removal of the lipoma. This patient checks in at the front desk and is guided back to the preoperative assessment area. There, the surgeon and anesthesiologist consent the patient for the procedure, start an IV, and ask the patient and his personal friend to say there goodbyes. Shortly thereafter, the patient is brought to the OR and sedated for the procedure. The case goes off without a hitch, and two hours later the patient is comfortably recovering from the effects of the sedative before being taken home by his friend. His wound heals well, the tissue removed during surgery is found to be benign, and he is formally released from the surgeon’s care at a follow-up appointment two weeks later.
This vignette represents a stylized “perfect” surgical encounter. All the various processes necessary to move from diagnosis of surgical disease to remediation of the condition proceed apace without incident. What happens in real life? In more than a few cases, one of the many steps above has a seemingly minor logistical hiccup that ultimately causes far too many resources to be devoted to this individual patient’s care. Sometimes these issues pass without the patient even aware of a problem. For example, it is not uncommon that a patient is brought to an operating room and sedated but the surgical team is not yet available to start the case. Other times, the patient’s entire memory of the encounter is shaped by the issue. It is not unheard of that a patient arrives for surgery and finds that because of paperwork errors he or she was not scheduled for surgery. In the best of cases, the patient has to wait a few hours before being squeezed in. Unfortunately, sometimes the patient is asked return at a later date for the procedure which of course requires the patient finding a new window in his busy schedule that can accomodate such a visit. Regardless of whether the patient is aware of the problem or not, these logistical errors greatly increase the resources and time needed to meet the healthcare needs of a population.
In the U.S., a seminal report by the Institute of Medicine (the profession’s pantheon) in 2001 radically accelerated the industry’s adoption of product quality and operations management practices from other industries. A practical example of this was many of the “cost containment” issues that were discussed — but largely not included — in the 2010 U.S. healthcare reform legislation. For example, the original legislation included powerful politically independent panels of experts (a la the 9/11 Commission) that would provide evidence-based – rather than expert-opinion – recommendations for the highly politicized but necessary changes to align Medicare reimbursement policies with cost-effective care.
Within surgery communities, there seems to be a rather large divide between institutions and programs that “get it” and are willing to spend the administrative resources to address these operational inefficiencies and those who fail to recognize the magnitude of these problems or see no solution from them.
The part of surgery’s newfound interest in “quality improvement” that is of greatest importance to me is exploring the incremental changes we can make to healthcare processes to prevent the small process problems noted above that ultimately weigh heavily on the costs of healthcare. I specifically am looking at using process mapping and decision analysis tools to question existing healthcare operations through a patient-centered perspective.
What we are trying to do in quality improvement is identify systems (e.g., patient management systems), products (e.g., mobile software), and services (e.g., nursing hotlines), that help us provide healthcare that is more comprehensive but also cheaper than existing practices. The burden that currently weighs on those of us working in this field is developing and testing these new methods of providing care in a way that convincingly demonstrates results with which we can act on.





